Tumor-Immune Interactions
Tumors are continuously interacting with the immune system.
- Various components of the immune system are involved in anti-tumor immunity, while others are involved in tumor progression
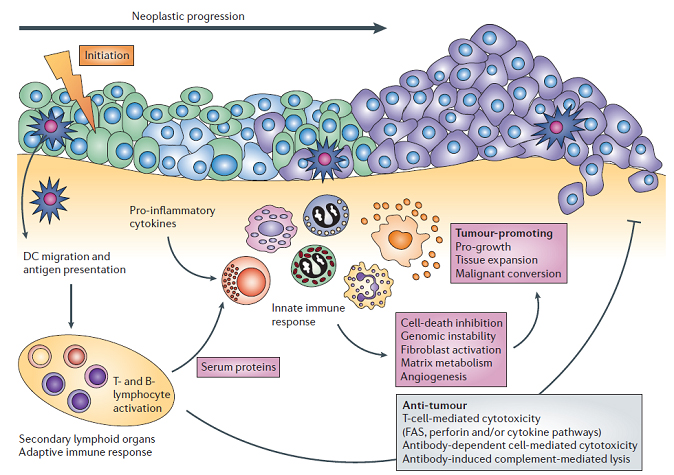 Image Caption
Image Caption
Used with permission from Macmillan Publishers Ltd: Nature Reviews Cancer, 6, 24-37, copyright 2006.
Immunoediting
- Together, the immune interactions involved in tumor surveillance and tumor progression have been collectively termed "immunoediting".
- Immunoediting is composed of 3 phases:
- Elimination = Immune cells recognize and destroy transformed cells to prevent the development of malignancy.
- Equilibrium= The tumor and immune system are in a dynamic balance where anti-tumor immunity controls tumor growth, but does not completely eliminate the tumor, so that tumor cells appear to be clinically dormant.
- Some tumor cells have evolved ways to evade detection and elimination by the immune system, such as upregulation of PD-L1.
- Escape = Genetic changes in the tumor or immunosuppressive mechanisms allow tumor cells to escape from control by the immune system and grow in an unrestricted manner.
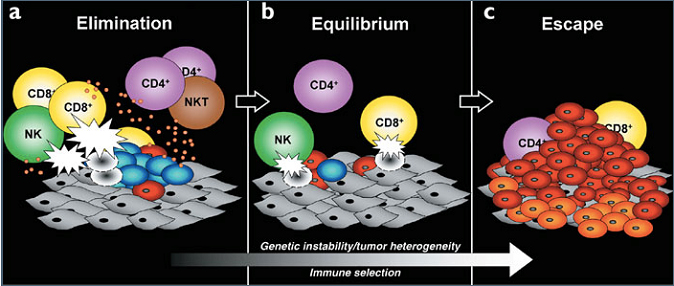
Used with permission from Macmillan Publishers Ltd: Nature Immunology, 3, 991-998, copyright 2002.
Mechanisms of Immune Evasion
- Downmodulation of tumor antigen presentation
- Many tumor cells downregulate MHC class I molecules to avoid detection by cytotoxic CD8+ T cells.
- Changes within the tumor microenvironment
- The tumor microenvironment can produce cytokines that skew macrophages from a tumor-eliminating M1 phenotype to a tumor-promoting M2 phenotype.
- Tumors produce inhibitory molecules such as indolamine 2,3-dioxygenase (IDO) to alter tryptophan catabolism and inhibit T cell responses.
- Tumors secrete molecules involved in remodeling of the extracellular matris in the tumor microenvironment, making it difficult for immune cells to access the tumor.
- Dysregulation of antigen presenting cells
- Dendritic cells within the tumor microenvironment often have low levels of proinflammatory cytokine production, costimulatory molecules and MHC class II expression.
- Induction of T cell tolerance
- Immature DCs in the tumor microenvironment cannot fully activate T cells, and instead induce anergic T cells that are unable to eliminate the tumor.
- Increased expression of co-inhibitory signals
- Tumors can increase expression of ligands for the inhibitory receptors, CTLA-4 and PD-1, which bind to B7-1/B7-2 or PD-L1/PD-L2. This decreases the amplitude of the antigen-specific T cell response.
- Induction of regulatory T cells
- Tumors secrete cytokines such as TGF-beta that induce T regulatory (iTreg) cells which suppress antigen-specific T cells.


