Sparking opportunity for an emerging leader in AI research
By Emily T. Connelly, MA, CRA, Speed of Light – The ASTRO Foundation
The research of Tafadzwa L. Chaunzwa, MD, MHS, at the intersection of artificial intelligence (AI), imaging, and radiation oncology is shaping new ways to understand and treat lung cancer. He leads the Advanced Computing and Oncology (ACO) Lab at Memorial Sloan Kettering Cancer Center (MSK), where he is a physician-scientist focused on treating lung cancer and related thoracic malignancies. When Dr. Chaunzwa was an ABR Holman resident at the Harvard Radiation Oncology Program, he received the James D. Cox Research Award from ASTRO’s foundation, then known as the Radiation Oncology Institute (ROI), to support his work applying AI-enabled quantitative imaging to personalize radiotherapy in the immunotherapy era and characterize radiotherapy-driven immune modulation in lung cancer. His journey illustrates how investments in early career researchers at pivotal moments can have lasting and significant impacts for both the individuals and the broader field.
Advancing the Science of Imaging and AI
Dr. Chaunzwa’s background in mathematics, biomedical engineering and data science led him to focus his research during medical school and residency on medical image analysis to extract features that can inform practice. With the grant from ROI, Dr. Chaunzwa led a project using quantitative imaging and foundation models to analyze CT scans in patients with non-small cell lung cancer to identify signatures that help predict their response to immunotherapy and radiotherapy. Foundation models are trained on large image datasets and then fine tuned for narrower clinical tasks, such as predicting tumor biology or characterizing host features including body composition. “It’s really a scalable and more efficient way of feature extraction,” Dr. Chaunzwa explained.Dr. Chaunzwa and his team combined the image analysis with clinical and demographic risk factors to develop frameworks that can predict response to immunotherapy and personalize radiotherapy. Selected results from this work were published in JAMA Oncology1 (June 2024) in the article “Body Composition in Advanced Non–Small Cell Lung Cancer Treated With Immunotherapy,” with additional findings on self-supervised learning for prognostication in this population presented at the 2023 World Conference on Lung Cancer.2
Although components of his research pipeline remain in active development, others are moving toward clinical translation. MSK’s strong infrastructure for tumor molecular profiling and investigator initiated trials has positioned the ACO Lab’s work for prospective validation — one of the most crucial steps toward making AI based tools available in real world practice. “We’re just excited to get our work out into the public domain,” Dr. Chaunzwa noted, emphasizing the importance of rigorous validation in advancing innovations toward regulatory approval.
A Transformational Milestone
For Dr. Chaunzwa, the grant he received from ROI marked an essential turning point. It was the first time he secured extramural funding as a principal investigator, which he described as a key milestone in his career. The Foundation’s investment allowed him to begin charting an independent scientific path, including hiring his first research assistant and purchasing equipment for research. Just as importantly, it prepared him to compete successfully for additional awards and grants from other organizations and the National Cancer Institute. He also laid the groundwork for establishing the ACO Lab, now 12 members strong, bringing together individuals from a broad range of academic and personal backgrounds. Dr. Chaunzwa shared, “One of my greatest aspirations is to be a thoughtful and supportive mentor as the members of my team chart their own paths. Their growth and contributions are central to everything we do.”Ultimately, the purpose of the team’s research is deeply patient centered. Understanding treatment sensitivity, predicting side effects, and identifying meaningful patterns in imaging all support a future in which lung cancer care is more personalized and effective. “This grant was a major catalyst for my research career. It gave me the chance to lead my own work, build a team, and accelerate discoveries that I hope will improve outcomes for people with lung cancer,” said Dr. Chaunzwa.
References
- Chaunzwa TL, Qian JM, Li Q, et al. Body Composition in Advanced Non-Small Cell Lung Cancer Treated With Immunotherapy. JAMA Oncol. 2024 Jun 1;10(6):773-783.
- Chaunzwa TL, Pai SL, Li Q, et al. P2.05-08 Immunotherapy Prognostication with Thoracic CT Contrastive Self-Supervised Learning in Patients with Non-Small Cell Lung Cancer. Journal of Thoracic Oncology. 2023 Nov;18(11):S31-S311.
Radiation Oncology's Next Top Model

Krishni Wijesooriya, PhD, of the University of Virginia sees value in both conventional statistical models and AI-charged versions.

For example, in her team’s analysis of immune suppression after SBRT for early-stage lung cancer, the modeling was traditional, with input variables extracted from patient records correlated with output variables from the same patients’ charts. The end product is an equation with dosimetric variables that can be validated on other retrospective data sets. And, hey, why stop there? How about proving the model in a prospective randomized trial? The UVA team have done this — a modified figure from one of their papers:1
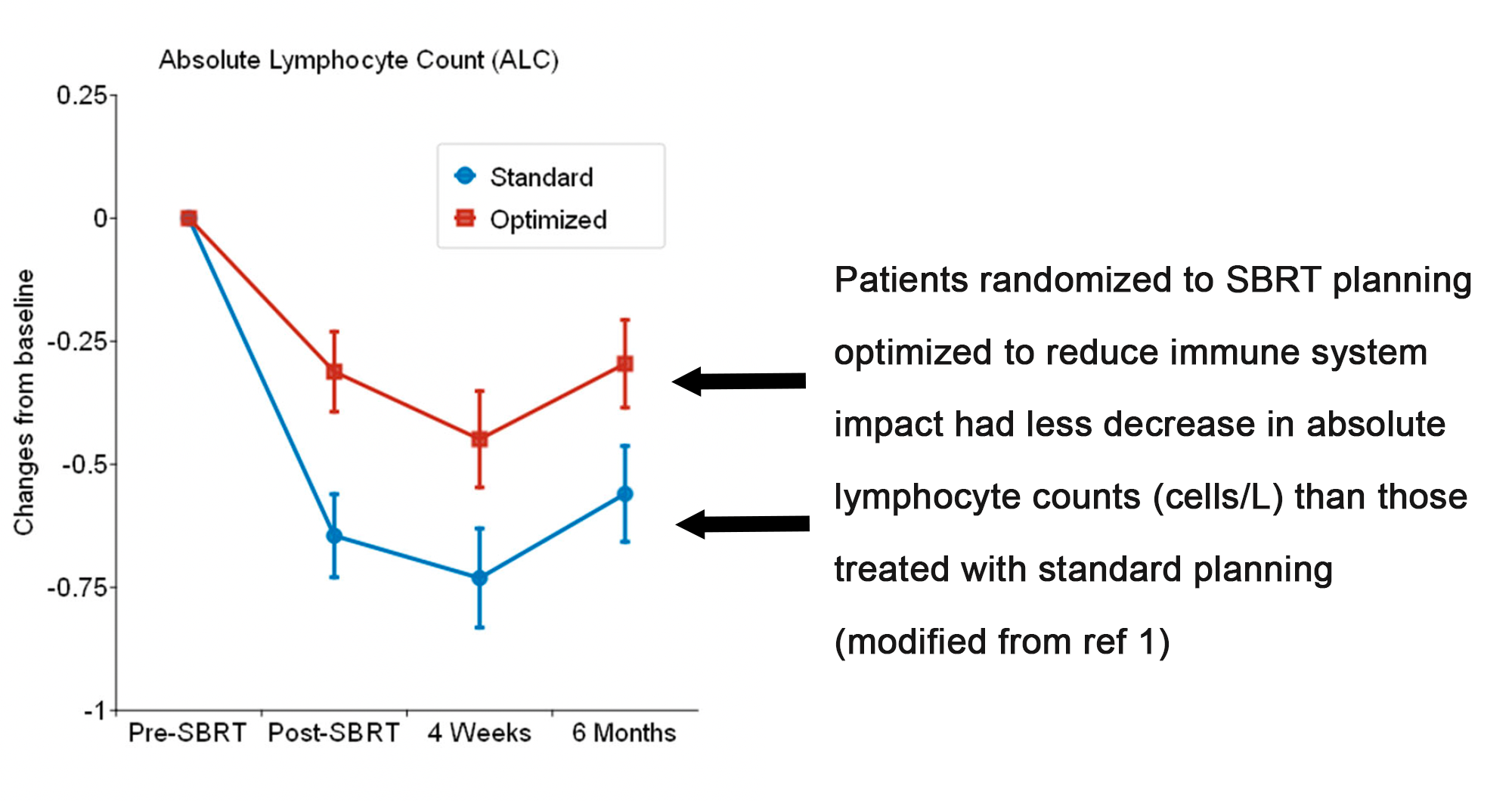
Contrast this study with the exercise Dr. Wijesooriya and team completed for a totally different study, a machine learning-based analysis of diagnostic CT scans to construct a predictive model to identify which scans revealed pre-invasive or invasive lung cancers.2 Here, the processing is more “black box,” insofar as the computer software extracts assorted radiomic features on its own and then generates predictions. Notably, though, there is an opportunity to make these models “smarter” in their predictions by intentionally augmenting the input data with image sets that have known malignant cancers.3

Looking ahead, taking a cue from both types of models, Dr. Wijesooriya wonders if the best path forward in this age of CPUs might actually be to think more…um…agriculturally?
“The next leap will come not merely from analyzing existing data but from actively generating new insights. The field should also farm new data,” says Dr. Wijesooriya. By which she means that we should develop hybrid models that combine AI-driven variables with input features that we can actually control, such as dose and schedule and adjuvant therapies. “If you have AI plus a conventional model hybrid, you know which parameters you can control and change.” And this, in turn, will allow us to produce new data that will refine our models in an augmentative manner, giving us a chance to move knowledge forward.
Ok, sure, looks good and arguably more stylish than workmanlike Conventional Model Man and more down-to-earth than elegant AI Model Man, but I think Farming Model Man’s scarf is over the top.
References
- Wijesooriya K, et al. First measurement: Proactive immune cell sparing in radiation therapy. medRxiv. 2025 Jan 6:2025-01.
- Huchthausen C, et al. Training Set Augmentation and Harmonization Enables Radiomic Models to Detect Early Onset of Lung Cancer. Preprint. Research Square. 2025.
- TL/DR: sorry, not possible to shorten this into a sentence or two. I’m afraid you have to read the paper.


