Cancer, the Immune System and Radiation Therapy
Early on, it was recognized that there is a dynamic interaction between cancer development, cancer progression and the patient's own immune system. Tissue changes early in the neoplastic process are detected by the host's innate immune system via immunosurveillance mechanisms including effector T cells, NK cells and macrophages which kill developing tumor cells. This leads to release of tumor-associated antigens which are taken up by dendritic cells, processed, and presented to CD8 and CD4 T cells. Additional proinflammatory signals activate T cells specific to the tumor, leading to an antitumor effect. This process is dynamic, whereby genetically unstable mutating cancer cells may continue to proliferate and be killed by T cells, but may ultimately develop mechanisms of escaping immune control.
Beyond the broadly cytotoxic effect of radiation therapy previously described, radiation therapy interacts with the tumor and immune system through a variety of mechanisms. Radiation therapy promotes the release of tumor neoantigens during cancer cell death in addition to stimulating immune adjuvant effects, engaging both innate and adaptive arms of the immune system and functioning like an in situ vaccine, generating tumor-specific T cells with local as well as potentially distant, systemiceffects. The key molecular signals generated by radiation-induced cell death that promote uptake of dying cancer cells by dendritic cells (antigen presenting cells integral to generation of the immune response), antigen cross-presentation and activation of the inflammasome collectively constitute immunogenic cell death. Moreover, radiation has complex effects on the tumor microenvironment, potentially allowing enhanced infiltration of activated T cells and helping to overcome the inhibition or exclusion of effector T cells.
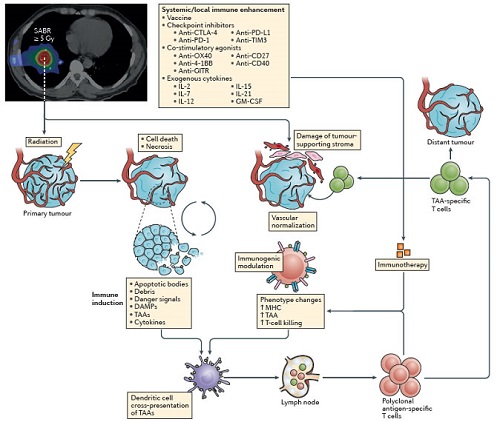
Used with permission from Macmillan Publishing Ltd: Nature Reviews Clinical Oncology, 13(8): 516-24, copyright 2016.
Despite this, a variety of factors pose barriers to the development of radiation-induced antitumor immunity. The limited availability of antigen-presenting cells, as well as immunosuppressive factors and cells (which may be enhanced by radiation itself) underlies the balance of factors at play in generating tumor-specific immunity by radiation, and potential barriers to inducing immunological memory and durable systemic response. Ongoing work to harness the proimmunogenic effects of radiation capitalize on the way in which radiation can complement the effects of immunotherapy to generate robust antitumor T-cell responses. A variety of trials in solid tumors are investigating the strategy of combining immunostimulatory signals with radiation, as well as the strategy of blocking negative regulators of dendritic cell and T cell function, even in poorly immunogenic tumor phenotypes.


